Among the existing energy storage technologies, lithium-ion batteries (LIBs) have unmatched energy density and versatility. Since their first commercialization, the growth in LIBs has been driven by portable devices. In recent years, however, large-scale electric vehicles and stationary applications have emerged.
Because LIB raw material deposits are unevenly distributed and prone to price fluctuations, these large-scale applications have put unprecedented pressure on the LIB value chain, resulting in the need for alternative energy storage chemistries. The sodium-ion battery (SIB or Na-ion battery) chemistry is one of the most promising “beyond-lithium” energy storage technologies.
Sodium-ion batteries are an emerging battery technology, on the cusp of commercialization, with promising cost, safety, sustainability, and performance benefits when compared to lithium-ion batteries. They can use widely available and inexpensive raw materials and existing lithium-ion production methods, promising rapid scalability.
SIBs are an attractive prospect in meeting global demand for carbon-neutral energy storage, where lifetime operational cost, not weight or volume, is the overriding factor. Increasingly sodium-ion batteries have characteristics comparable to lithium iron phosphate (LFP) batteries, suggesting that even automotive applications are possible.
SIBs have the same fundamental working principle as LIBs but rely on sodium rather than lithium as mobile cations. Unlike lithium, sodium does not electrochemically alloy with aluminum at room temperature. Thus, the copper current collector on the anode can be replaced by cheaper aluminum; it not only lowers the SIB costs, but also reduces the transportation risks, as SIBs can be transported completely discharged at 0V.
Hard carbon is typically used as the anode active material instead of graphite, as crystalline graphite has poor storage capabilities for sodium ions. Various cathode chemistries based on layered transition metal oxides, polyanionic compounds, and Prussian Blue Analogues can be used. Electrolytes and separators, as well as the positive current collectors, are similar to LIBs, except for the use of sodium salts in the electrolyte.
What Markets Exist for Sodium-Ion Batteries?
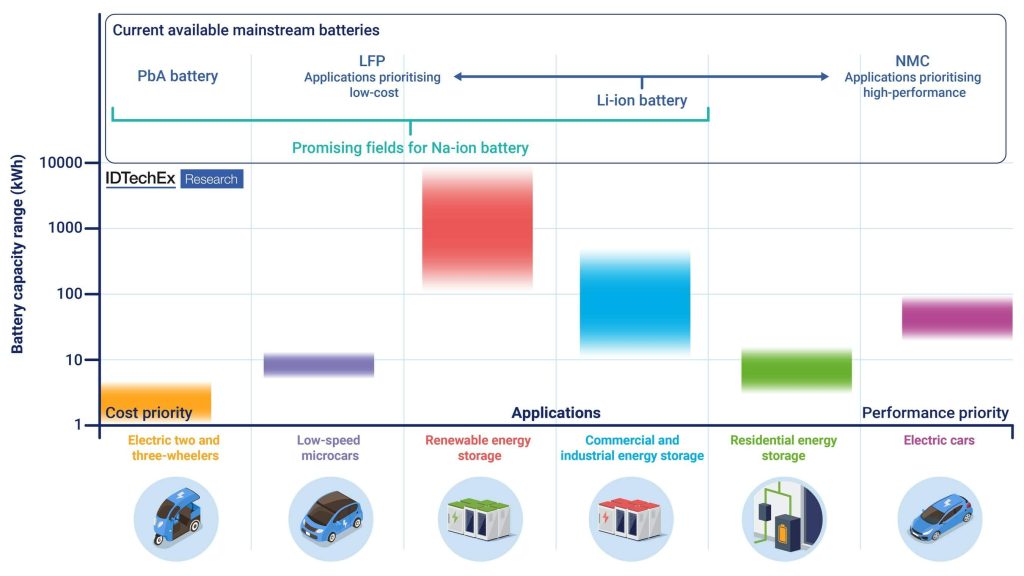
Although Na-ion technology mimics Li-ion with similar types of electrodes and electrolytes, Na is three times heavier than Li and has a redox potential 300mV lower, which inherently reduces the energy density of Na-ion technology by at least ∼30% compared to Li-ion. Therefore, the usage of Na-ion technology alone in applications requiring high energy density, such as battery electric cars, is partly eliminated. However, in applications where energy density is not as critical, for example stationary energy storage, electric-two wheelers, and electric microcars, Na-ion batteries can be ideal due to their power, safety, and cost characteristics.
Industrial Developments
Most Na-ion companies are based in China. The Chinese government is supporting the development of the country’s Na-ion battery industry with a view to securing a stable supply of batteries and taking the lead in the development of next-generation batteries. IDTechEx research finds nearly 20 companies working on sodium-ion battery technology in China alone. This is reflected by the patent activity being highest in the region as well. Despite lingering uncertainties about market prospects, companies in Western countries are making moves to enter the business, seeing the future potential of Na-ion batteries.
